A) The carbon in the alcohol is sp2 hybridized and has a higher percent s-character that lengthens the C-O bond in the alcohol.
B) The carbon in the carboxylic acid is sp3 hybridized and has a lower percent s-character that shortens the C-O bond in the carboxylic acid.
C) The carbon in the carboxylic acid is sp hybridized and has a higher percent s-character that shortens the C-O bond in the carboxylic acid.
D) The carbon in the carboxylic acid is sp2 hybridized and has a higher percent s-character that shortens the C-O bond in the carboxylic acid.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would happen if a mixture of benzoic acid and cyclohexanol dissolved in CH2Cl2 is treated with aqueous NaOH solution?
A) Benzoic acid would remain in the CH2Cl2 layer,and cyclohexanol would dissolve in the aqueous layer.
B) Benzoic acid would dissolve in the aqueous layer while cyclohexanol would remain in the CH2Cl2 layer.
C) The salt of benzoic acid would dissolve in the aqueous layer while cyclohexanol would remain in the CH2Cl2 layer.
D) The salt of benzoic acid would remain in the CH2Cl2 layer while cyclohexanol would dissolve in the aqueous layer.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 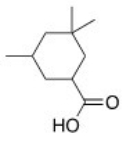
A) 3,5,5-Trimethylcyclohexanecarboxylic acid
B) 3,3,5-Trimethylcyclohexanecarboxylic acid
C) 3,3,5-Trimethylcyclohexanoic acid
D) 3,5,5-Trimethylcyclohexanoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is least basic? 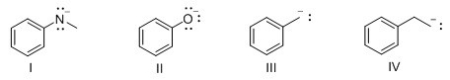
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What physical property and reaction type are used by extraction as useful techniques to separate and purify mixtures of compounds?
A) Physical property = solubility differences; reaction type = acid-base reaction
B) Physical property = boiling point; reaction type = acid-base reaction
C) Physical property = solubility differences; reaction type = oxidation-reduction
D) Physical property = density; reaction type = oxidation-reduction
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the following compound? 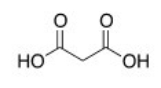
A) Propanedioic acid
B) 1,3-Propanedicarboxylic acid
C) Malonic acid
D) Succinic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 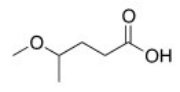
A) 2-Methoxypentanoic acid
B) 2-Methoxybutanoic acid
C) 4-Methoxybutanoic acid
D) 4-Methoxypentanoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As applied to the chemistry of amino acids,what is the definition for the isoelectric point?
A) The pH at which the amino acid exists primarily in its acidic form.
B) The pH at which the amino acid exists primarily in its basic form.
C) The pH at which the amino acid exists as a mixture of isomers.
D) The pH at which the amino acid exists primarily in its neutral form.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the presence of strong acids,which of the oxygen atoms on the carboxyl group is preferentially protonated and why?
A) Protonation occurs at the carbonyl oxygen because the resulting conjugate acid is stabilized by resonance.
B) Protonation occurs at the carbonyl oxygen because the resulting conjugate base is stabilized by the inductive effect.
C) Protonation occurs at the hydroxyl oxygen because the resulting conjugate acid is stabilized by resonance.
D) Protonation occurs at the hydroxyl oxygen because the resulting conjugate base is stabilized by the inductive effect.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity,putting the least acidic first. 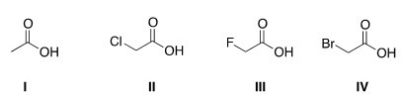
A) III < II < IV < I
B) III < IV < II < I
C) I < IV < II < III
D) I < II < III < IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 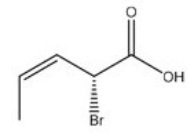
A) (E,R) -2-bromo-3-pentenoic acid
B) (Z,R) -2-bromo-3-pentenoic acid
C) (E,S) -2-bromo-3-pentenoic acid
D) (Z,S) -2-bromo-3-pentenoic acid
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 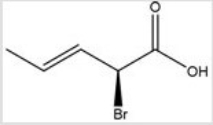
A) (E,R) -2-bromo-3-pentenoic acid
B) (Z,R) -2-bromo-3-pentenoic acid
C) (E,S) -2-bromo-3-pentenoic acid
D) (Z,S) -2-bromo-3-pentenoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity,putting the least acidic first. 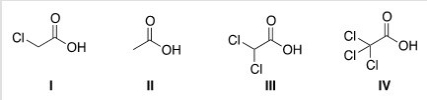
A) II < III < I < IV
B) II < I < III < IV
C) IV < III < I < II
D) IV < I < II < III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 53 of 53
Related Exams