B) False
Correct Answer

verified
Correct Answer
verified
Essay
Identify the acid, base, conjugate acid, conjugate base in the following reaction.
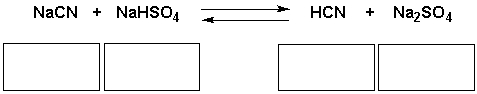
Correct Answer

verified
base, acid...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which substances are Lewis bases?
)
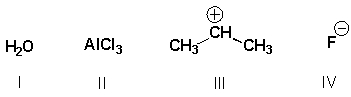
A) I, II
B) I, III
C) III, IV
D) I, IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the order of increasing acid strength of the following compounds (least first) ?
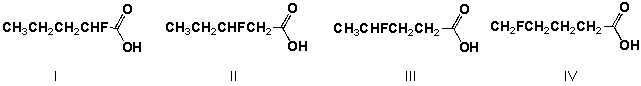
A) I, III, II, IV
B) IV, III, II, I
C) II, I, III, IV
D) IV, III, I, II
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Complete the following reaction.
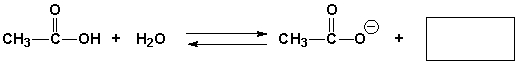
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the conjugate bases in the following reactions.
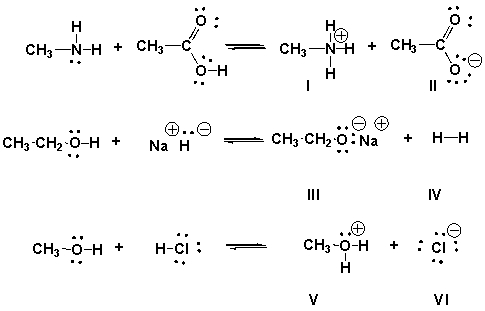
A) II, III, VI
B) I, IV, V
C) I, III, V
D) II, IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Complete the following reaction.
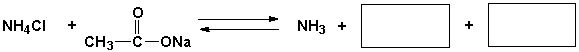
Correct Answer

verified
Correct Answer
verified
True/False
Water acts as a Brønsted-Lowry base in the following reaction.

B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
The weaker the acid, the ________________ the conjugate base.
Correct Answer

verified
Correct Answer
verified
Short Answer
Complete the following reaction.
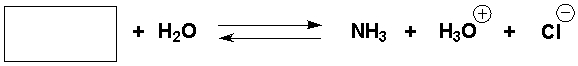
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the role of diethyl ether in the following reaction?

A) Lewis acid
B) Lewis base
C) Brønsted acid
D) Brønsted base
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which are acid-base reactions according to the Brønsted-Lowry theory?
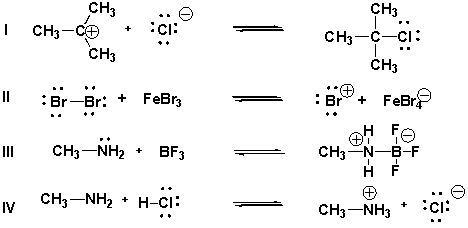
A) IV
B) I, III, IV
C) II, III
D) I, IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The equilibrium constant will be greater than 1.0 for the following reaction

B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the Arrhenius acids: I. HCl II. NaOH III. HNO3 IV. Ca(OH) 2
A) I, II
B) I, III, IV
C) II, IV
D) I, III
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the conjugate acids in the following reactions.
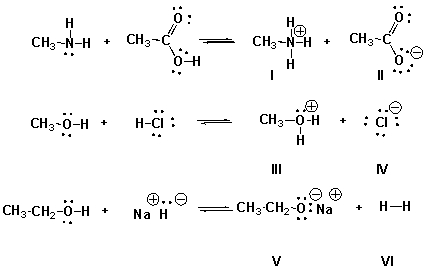
A) I, IV, VI
B) I, III, VI
C) II, IV, V
D) I, III, V
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the stronger acid in the following reaction if the equilibrium constant is approximately 108.
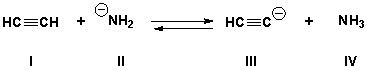
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the bonds in order of increasing acidity (least to most) .
A) II, III, IV, I
B) III, I, II, IV
C) I, IV, II, III
D) IV, III, II, I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Brønsted-Lowry acids accept protons when reacting.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The stronger acid has the larger (more positive) pKa.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Strong acids have weak conjugate bases.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 47
Related Exams