A) H+ and I -
B) H+ and HOI
C) HOI and OH-
D) H+ only
E) H2O and OH-
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 25°C, by what factor is the reaction rate increased by a catalyst that reduces the activation energy of the reaction by 1.00 kJ/mol?
A) 1.63
B) 123
C) 1.04
D) 1.50
E) 2.53
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The thermal decomposition of acetaldehyde, CH3CHO CH4 + CO, is a second-order reaction.The following data were obtained at 518°C.
 Based on the data given, what is the half-life for the disappearance of acetaldehyde?
Based on the data given, what is the half-life for the disappearance of acetaldehyde?
A) 1.5 × 105 s
B) 410 s
C) 5.4 × 107 s
D) 520 s
E) 305 s
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The activation energy for the following reaction is 60.kJ/mol. Sn2+ + 2Co3+ Sn4+ + 2Co2+ By what factor (how many times) will the rate constant increase when the temperature is raised from 10°C to 28°C?
A) 1.002
B) 4.6
C) 5.6
D) 2.8
E) 696
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
The rate constant for a certain first-order reaction is 0.40/min.What is the initial rate in mole/L·min, if the initial concentration of the compound involved is 0.50 mol/L?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following data to determine the rate law for the reaction shown below. 2NO + H2 N2O + H2O ![Use the following data to determine the rate law for the reaction shown below. 2NO + H<sub>2</sub> \rarr N<sub>2</sub>O + H<sub>2</sub>O A) rate = k[NO] B) rate = k[NO]<sup>2</sup> C) rate = k[NO][H<sub>2</sub>] D) rate = k[NO]<sup>2</sup>[H<sub>2</sub>] E) rate = k[NO]<sup>2</sup>[H<sub>2</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f16_b8fc_a2ab_b9a22cd78793_TB3246_00.jpg)
A) rate = k[NO]
B) rate = k[NO]2
C) rate = k[NO][H2]
D) rate = k[NO]2[H2]
E) rate = k[NO]2[H2]2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the slope of an Arrhenius plot for the following reaction?
2NOCl 2NO + Cl2

A) 8.18 × 10-2 K
B) 5.06 × 10-2 K
C) -1.22 × 104 K
D) -1.96 × 104 K
E) not enough information to calculate the slope
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A city's water supply is contaminated with a toxin at a concentration of 0.63 mg/L.For the water to be safe for drinking, the concentration of this toxin must be below 1.5 x 10-3 mg/L.Fortunately, this toxin decomposes to a safe mixture of products by first-order kinetics with a rate constant of 0.27 day-1.How long will it take for the water to be safe to drink?
A) 2.2 days
B) 2.6 days
C) 20.days
D) 22 days
E) 27 days
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Concerning the rate law, Rate = k[A]2[B], what are appropriate units for the rate constant k?
A) s-1
B) M-1s-1
C) M-2s-1
D) M/s
E) M2/s
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction C6H14(g) C6H6(g) + 4H2(g) , P(H2) / t was found to be 2.5 × 10-2 atm/s, where P(H2) is the change in pressure of hydrogen.Determine P(C6H14) / t for this reaction at the same time.
A) 2.5 × 10-2 atm/s
B) -6.2 × 10-3 atm/s
C) -2.5 × 10-2 atm/s
D) 0.10 atm/s
E) 6.2 × 10-3 atm/s
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Nitric acid is formed by the gas-phase hydrolysis of N2O5.For the reaction N2O5 + H2O 2HNO3, Ea(forward)= 15 kJ/mol and Ea(reverse)= 51 kJ/mol.Calculate Hrxn.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate law for the reaction 2NO2 + O3 N2O5 + O2 is rate = k[NO2][O3].Which one of the following mechanisms is consistent with this rate law?
A) NO2 + NO2 N2O4 (fast) N2O4 + O3 N2O5 + O2 (slow)
B) NO2 + O3 NO5 (fast) NO5 + NO5 N2O5 + 5/2O2 (slow)
C) NO2 + O3 NO3 + O2 (slow) NO3 + NO2 N2O5 (fast)
D) NO2 + NO2 N2O2 + O2 (slow) N2O2 + O3 N2O5 (fast)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following mechanism has been suggested for the reaction:
H2O2 + 2H+ + 2I - I2 + 2H2O
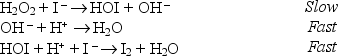 Identify the molecularity of the rate determining step
Identify the molecularity of the rate determining step
A) unimolecular
B) bimolecular
C) termolecular
D) unimolecular and bimolecular
E) bimolecular and termolecular
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following statement: A catalyst
A) increases the activation energy.
B) alters the reaction mechanism.
C) increases the average kinetic energy of the reactants.
D) increases the concentration of reactants.
E) increases the collision frequency of reactant molecules.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction A + 3B 2C, the rate of disappearance of B given by ( [B]/ t) may also be expressed as
A) ( [B]/ t = - (1/3) [A]/ t)
B) ( [B]/ t = - 3 [A]/ t)
C) ( [B]/ t = 3 [A]/ t)
D) ( [B]/ t = (1/3) [A]/ t)
E) ( [B]/ t = - [A]/ t)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If Ea for a certain biological reaction is 50.kJ/mol, by what factor (how many times) will the rate of this reaction increase when body temperature increases from 37°C (normal) to 40°C (fever) ?
A) 1.15
B) 1.20
C) 2.0 × 105
D) 1.0002
E) 2.0
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chlorine dioxide reacts in basic water to form chlorite and chlorate according to the following chemical equation: 2ClO2(aq) + 2OH-(aq) ClO2-(aq) + ClO3-(aq) + H2O(l) Under a certain set of conditions, the initial rate of disappearance of chlorine dioxide was determined to be 2.30 x 10-1 M/s.What is the initial rate of appearance of chlorite ion under those same conditions?
A) 5.75 x 10-2 M/s
B) 1.15 x 10-1 M/s
C) 2.30 x 10-1 M/s
D) 4.60 x 10-1 M/s
E) 9.20 x 10-1 M/s
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction whose rate law is rate = k[X], a plot of which of the following is a straight line?
A) [X] versus time
B) ln [X] versus time
C) 1/[X] versus time
D) [X] versus 1/time
E) ln [X] versus 1/time
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium ion (NH4+) reacts with nitrite ion (NO2-) to yield nitrogen gas and liquid water.The following initial rates of reaction have been measured for the given reactant concentrations. ![Ammonium ion (NH<sub>4</sub><sup>+</sup>) reacts with nitrite ion (NO<sub>2</sub><sup>-</sup>) to yield nitrogen gas and liquid water.The following initial rates of reaction have been measured for the given reactant concentrations. Which of the following is the rate law (rate equation) for this reaction? A) rate = k [NH<sub>4</sub><sup>+</sup>] [NO<sub>2</sub><sup>-</sup>]<sup>4</sup> B) rate = k [NH<sub>4</sub><sup>+</sup>] [NO<sub>2</sub><sup>-</sup>] C) rate = k [NH<sub>4</sub><sup>+</sup>] [NO<sub>2</sub><sup>-</sup>]<sup>2</sup> D) rate = k [NH<sub>4</sub><sup>+</sup>]<sup>2</sup> [NO<sub>2</sub><sup>-</sup>] E) rate = k [NH<sub>4</sub><sup>+</sup>]<sup>1/2</sup> [NO<sub>2</sub><sup>-</sup>]<sup>1/4</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f17_2e30_a2ab_09f7cd5b313e_TB3246_00.jpg) Which of the following is the rate law (rate equation) for this reaction?
Which of the following is the rate law (rate equation) for this reaction?
A) rate = k [NH4+] [NO2-]4
B) rate = k [NH4+] [NO2-]
C) rate = k [NH4+] [NO2-]2
D) rate = k [NH4+]2 [NO2-]
E) rate = k [NH4+]1/2 [NO2-]1/4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Appropriate units for a first-order rate constant are
A) M/s
B) 1/M·s
C) 1/s
D) 1/M2·s
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 130
Related Exams